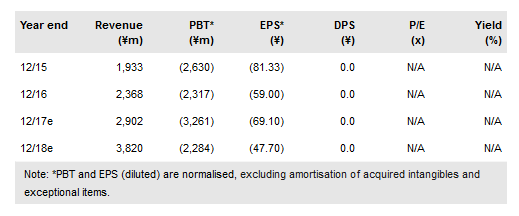
During 2017 we expect updates on all three of SymBio’s main assets. We expect Treakisym sales growth via partner Eisai Co., Ltd. (T:4523) following approvals during 2016 in new indications, which could more than double the current levels (¥4.7bn). For pipeline assets IONSYS and rigosertib, data from ongoing trials could become available during H217, which will shape the future development pathways. We also expect Symbiox Investment & Trading Co Ltd (BO:SYMB) to remain focused on in-licensing further opportunities to complement its existing pipeline. This could help drive future operating leverage if SymBio evolves into a commercial entity, possibly from 2020. We value SymBio at ¥19.7bn.
On track to maximise Treakisym’s potential
During 2016 SymBio received additional approvals for Treakisym in chronic lymphocytic leukaemia (CLL) and first-line low-grade non-Hodgkin’s lymphoma (NHL) and mantle cell lymphoma (MCL). These represent patient markets in Japan of around 7,800, compared to around 4,700 patients with relapsed or refractory (r/r) low-grade NHL/MCL, where sales have been generated to date (¥4,200m in 2016) via partner Eisai. Hence, we believe these additional indications could more than double current Treakisym sales and we expect an uplift during 2017.
To read the entire report Please click on the pdf File Below