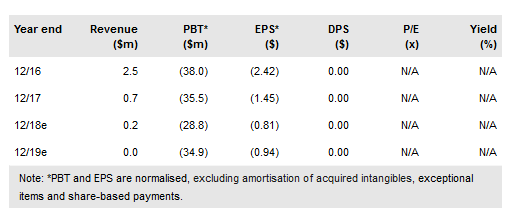
Sunesis Pharmaceuticals Inc (NASDAQ:SNSS) released its abstracts for the upcoming American Society of Hematology (ASH) meeting, which included an update on the company’s ongoing Phase Ib/II trial of vecabrutinib in hematologic cancers. The study is still in the dosing portion of the trial on the 50mg arm, but the data to date showed a safety and tolerability profile in line with expectations. The company will provide a complete update of the trial progress at ASH.
The 50mg arm is continuing to enrol
Vecabrutinib is in the dosing portion of a dose escalation/expansion study in patients with chronic lymphocytic leukemia and other hematologic malignancies. The study hit a delay earlier this year when it encountered a dose-limiting toxicity (DLT) that triggered an expansion of the 50mg cohort. In the abstract, the company revealed that some of these patients progressed before completion of their first course, necessitating further enrolment. The 50mg dose is below the threshold of clinical activity, so it is simply an unfortunate risk they have encountered.
To read the entire report Please click on the pdf File Below..