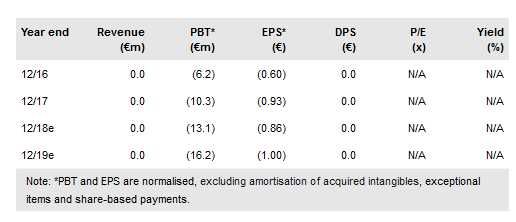
Quantum Genomics (PA:ALQGC) released data from the Phase IIb NEW-HOPE trial, which strongly suggests that firibistat is an efficacious, safe drug with a differentiated mechanism that will address a very large patient population. After eight weeks of treatment, patients saw a statistically significant reduction from baseline (p<0.0001) in systolic automated office blood pressure (AOBP) of 9.7mmHg. Importantly there was no oedema that was seen with some of the other major classes of hypertension treatments.
Efficacy similar to major blockbusters
While it is hard to compare across trials as there are differences in clinical trial design, the NEW-HOPE data is fairly similar to the eight-week treatment data for Diovan (valsartan), which had peak sales of $6bn in 2010 (although this also includes sales in heart failure patients) and had shown 6–9mmHg reductions in systolic blood pressure in their eight-week hypertension studies.
To read the entire report Please click on the pdf File Below..