IMP321 set to progress into Phase IIb study
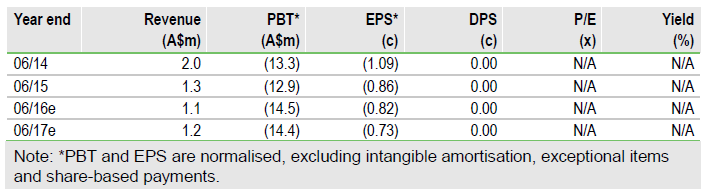
Prima Biomed Ltd (AX:PRR) is well positioned to push forward with the clinical development of its promising LAG3 programme following an A$25m fund raising and encouraging scientific advice from the EMA for its upcoming Phase IIb trial of IMP321 in metastatic breast cancer. Novartis AG (N:NVS) and GlaxoSmithKline PLC (N:GSK) have recently commenced clinical trials of partnered LAG3 programmes, providing additional validation for the LAG3 technology that Prima obtained with the 2014 acquisition of Immutep. We lift our valuation to A$271m (vs A$199m) with the inclusion of the Novartis LAG3 programme now that it has entered the clinic, and a melanoma indication for IMP321.
Novartis moves IMP701 programme into the clinic
Novartis has initiated a Phase I trial of LAG525, its humanised version of Prima’s IMP701 antagonist anti-LAG-3 antibody, triggering a modest milestone payment. Novartis will trial LAG525 in cancer patients both as a single agent and in combination with its immune checkpoint inhibitor PDR001, underscoring the potential benefits that it sees from targeting LAG3.
To Read the Entire Report Please Click on the pdf File Below