The growing US Zubsolv franchise continues to drive Orexo’s quarterly performance. Total Q3 revenue growth of c 30% puts Orexo in an attractive position with potential business development partners. Orexo’s Q318 results were the first since Zubsolv’s exclusivity was strengthened for another 13 years in the US, so with Zubsolv sales growing by 1.2% q-o-q in Q3, against the backdrop of a 0.3% decline in the buprenorphine/naloxone market volumes, and continued material CoGS improvements, Orexo AB (ST:ORX) is moving from strength to strength.
Q318 results
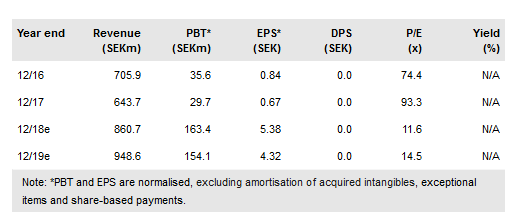
Total net revenues in Q318 were SEK216.6m, a 30.3% y-o-y increase, slightly below our estimates due to lower royalties from Abstral and Edluar. The main event – Zubsolv US revenue – was SEK165.4m (up 36.6% y-o-y in SEK terms, 24.3% in local currency), SEK4.4m above our estimates. The manufacturing efficiency programme reduced CoGS to SEK42.4m; lower than our recently reduced Q3 estimates of SEK49.6m and including a SEK1.5m stability testing cost. Gross cash balance at end-Q3 was SEK516.6m (SEK494.8m at end-Q218). Selling expenses of SEK51.5m were one-third lower than our estimates (despite integrating the US contract salesforce), as were R&D expenses, which, at SEK37.5m, were c 18% below our estimates. Administrative expenses of SEK50.8m were 60% higher than our estimates due to the SEK31.2m legal expense for the IP litigations. We have made adjustments accordingly but keep to FY18 opex guidance of c SEK500m. Profitability was enhanced by a SEK31.8m tax adjustment, which we are assuming was not a one-off and in Q318, it more than offset the IP litigation costs.
To read the entire report Please click on the pdf File Below.