Stock market today: S&P 500 slides on economic worries; Tesla hits closing record
Nanobiotix (PA:NANOB) has made progress with NBTXR3 as a standalone agent to enhance radiation therapy and now has clinical data from three cancers demonstrating consistent safety, feasibility and transferability of effect across different indications. Nanobiotix has also released preclinical results demonstrating NBTXR3’s ability to enhance the immunogenicity of various cancers, which is the cornerstone idea behind the immuno-oncology (IO) products.
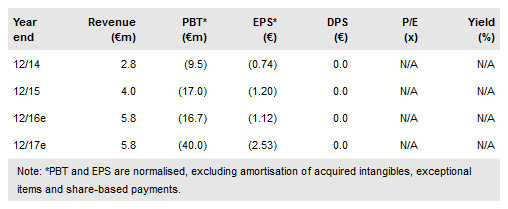
We expect an eventful 2017, with several clinical trials reporting results and a potential CE mark approval in mid-2017 for use in soft tissue sarcoma (STS). Our valuation has increased to €738m.
Application for CE mark filed; good H&N cancer data
In August 2016, Nanobiotix filed a CE mark application for NBTXR3. The data package was based on existing evidence obtained in clinical trials so far, including the most advanced registration Phase II/III trial in STS, which is currently ongoing. Evaluation started in September and should last at least nine months. According to the latest update, 115 patients have been randomized into the Phase II/III STS trial and the interim analysis readout is expected in the coming months.
In July 2016, Nanobiotix also presented clinical data from Phase I/II H&N cancer, indicating promising signs of tumour response in all treated patients. This was a second indication with clinical data and has demonstrated that NBTXR3’s effect seems to be transferable to other solid tumours given its mechanistic mode of action. More recently, similar positive results were announced from Phase I/II in liver cancers.
To read the entire report please click on the pdf file below