U.S. consumer inflation rose by less than expected in November; CPI gained 2.7%
At the American Society of Hematology (ASH) annual meeting, compelling early combination data on MOR208 and MOR202 were presented. MOR208 with lenalidomide in previously untreated chronic lymphocytic leukemia (CLL) patients demonstrated a 100% (four/four) response rate in evaluable patients. In the relapsed/refractory CLL population, the combination demonstrated a 43% (3/7) response rate. MOR202 in combination with lenalidomide or pomalidomide demonstrated a 50% (three/six) response rate in relapsed/refractory multiple myeloma (MM) patients.
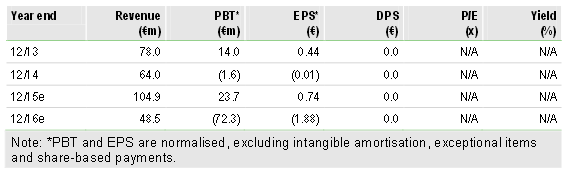
MOR208 continues to progress
At ASH, MOR208 data in non-Hodgkin’s lymphoma (NHL) and CLL were presented. While the NHL poster was a minor update, the CLL combination data were new and promising, despite the small size of the study. Morphosys AG O.N. (DE:MORG) plans to initiate an 80-patient Phase II in combination with lenalidomide in second line, relapsed/refractory DLBCL patients in Q415. It also plans to initiate a 120-patient Phase II trial in CLL in combination with idelalisib in BTKi-failures in Q116.
To read the entire report Please click on the pdf File Below