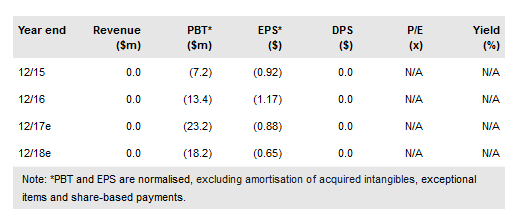
Intec Pharma's (NASDAQ:NTEC) clinical programmes to validate the utility of its unique drug delivery platform, the accordion pill (AP), are ongoing. The company announced with its Q317 report that patient enrolment for the Phase III AP-CDLD (carbidopa and levodopa) in Parkinson’s has been increased to 420 patients (from 328). We now expect full enrolment in Q318. Additionally, Intec is redesigning AP-CBD/THC (cannabidiol and tetrahydrocannabinol) for a second Phase I trial and the indication is not yet specified.
AP-CDLD enrolment upsized and trial delayed
Intec has announced that it is expanding target enrolment for the Phase III AP-CDLD trial to 420 patients (after downsizing enrolment to 328 from 460 patients), due to higher than expected dropout rates associated with endoscopy procedures prior to randomization and open-label Sinemet titration that comes before AP exposure. The company now expects completion of enrolment in Q318 (previously Q417). Provided that patients are followed for 26 weeks, we expect top-line data in mid-2019.
To read the entire report Please click on the pdf File Below: