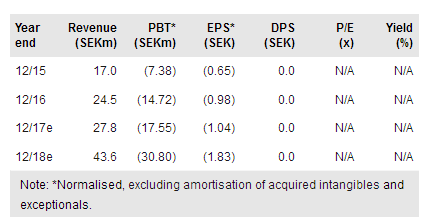
Immunovia publ AB (ST:IMMNOV) is on track to start commercialising its PanCan-d test for the early detection of pancreatic cancer in high-risk patients in 2018. To this end, it has established its US headquarters in Boston, which includes a laboratory regulated via the Clinical Laboratory Improvement Amendments (CLIA) programme. It has also identified a new important opportunity for the PanCan-d test in the early pancreatic cancer symptoms group, has announced further positive data on the differentiation of autoimmune diseases, and continues the preparations for a retrospective study and a biomarkers study in the diabetic population. Our valuation is SEK157/sh.
First PanCan-d sales on track for 2018
As previously guided, Immunovia plans to start out-of-pocket sales in 2018 in the US (as a CLIA-accredited laboratory developed test) and the EU (CE mark). It will seek reimbursement after the PANFAM-1 study, a three-year prospective clinical trial in 1,000 high-risk individuals with results expected in 2019. Immunovia aims to have PanCan-d included in surveillance programmes in familial pancreatic cancer and we estimate US and EU sales could represent a SEK2bn opportunity.
To read the entire report Please click on the pdf File Below: