Clarity on resminostat strategy
4 SC AG (VSCG.F) has clarified and simplified its development strategy for resminostat in first-line liver cancer. Having previously considered an adaptive Phase II/III study, requiring a partner and/or significant investment today, the company now plans to conduct a Phase II study with the aim of confirming the drug’s efficacy and fully validating the proposed biomarker, ZFP64. This could deliver a more compelling partnering and development case for pivotal studies. We welcome this revision, although funding for the Phase II study will need to be secured and a longer clinical timeline (launch in 2021 vs 2019) reduces our overall valuation to €105m, or €2.07 per share.
Encouraging data…
The 57-patient, open-label, Phase IIa SHELTER study produced encouraging progression-free survival (PFS) and overall survival (OS) rates for resminostat in second-line liver cancer. The survival benefit appeared to be more pronounced in patients with high levels of the blood-based biomarker ZFP64. However, the ZFP64-correlated benefit was identified in a post-hoc analysis (ie not pre-defined in the trial design) and the patient numbers were relatively small.
…but further validation required
The previously proposed adaptive Phase II/III pivotal study design, with ZFP64 an integral part of patient stratification/selection, could have been achievable, but the hurdles to gaining regulatory approval and securing the finance and/or a pharma partner for this plan were probably too high. We therefore welcome the more straightforward intention to conduct a Phase II study, to provide further evidence to support resminostat’s benefit in combination with sorafenib (Nexavar) and validate ZFP64 (and potentially others) as a biomarker to inform the pivotal study design.
Creating a more valuable asset
Generating this Phase II data should increase resminostat’s value/attractiveness to prospective partners, although the size, scope and timelines are to be determined. 4SC will now ascertain support for the plan with regulators and potential investors/partners and expects to submit the study protocol by the end of 2014.
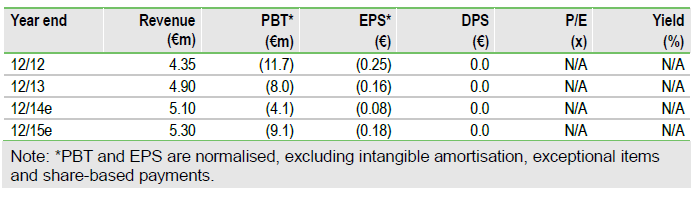
Valuation: Reduced to €105m (€2.07 per share)
Opting for stepwise Phase II then Phase III studies (not Phase II/III) pushes back the expected launch for resminostat from 2019 to 2021. This reduces our valuation to €105m (vs €122m) or €2.07/share (€2.41). However, we view this as a more realistic approach and are hopeful that the required funds (c €15m) can be secured.
To Read the Entire Report Please Click on the pdf File Below