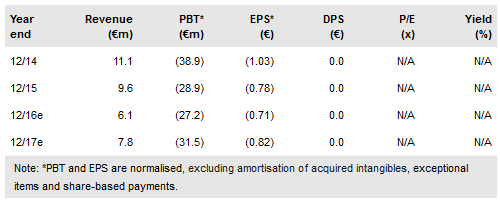
During Q316 Transgene (PA:TRNG) announced a collaboration agreement with Pfizer (NYSE:PFE) and Merck (NYSE:MRK) to develop TG4001 in combination with avelumab in a Phase I/II trial, the recruitment of the first patient in the multiple-dose cohort of Phase I/Ib of TG4001 in HBV patients and preclinical data from its vaccinia technology platform. The company plans to raise €48.1m (gross) through a rights issue that will fund operations to the end of 2018. Cash and equivalents at end Q316 amount to €25.4m. We value Transgene at €161.5m or €4.2/share.
Rights issue to fund strategy into first readouts
Transgene announced a rights issue for a planned gross amount of €48.1m. The subscription period will end on 4 November. The company will use funds to continue with its strategy of combining key pipeline assets with immune checkpoint inhibitors. In total, seven clinical trials are due to start before end 2017, with the first readouts expected in H217. With cash and equivalents of €25.4m at end Q316, the company guides FY16 cash burn at €35m.
To read the entire report Please click on the pdf File Below