FDA accepts filing of Beleodaq
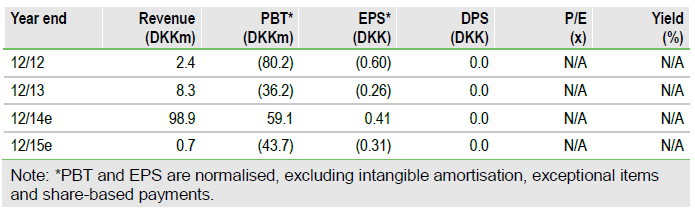
TopoTarget's, (TOPO) belinostat, now with the brand name Beleodaq, could be approved on 9 August 2014 (PDUFA date). The FDA has accepted the product filing and granted priority review. Topotarget will receive milestone payments worth c $17.8m from its partner Spectrum, and could receive a further $25m for product approval. We continue to be confident that belinostat will be approved, due to data from the BELIEF trial. In the meantime, Topotarget should disclose its broader development plans for belinostat in H114. We increase our valuation to DKK1,434m.
Filing accepted, PDUFA date is 9 August 2014
The FDA has accepted the filing of belinostat (or Beleodaq) for relapsed or refractory peripheral T-cell lymphoma (r/r PTCL), triggering milestone payments from Spectrum Pharmaceuticals of $10m in cash and 1m of its shares (worth c $7.8m). The PDUFA date is 9 August 2014, following the FDA’s decision to grant priority review. If belinostat is approved in August, Spectrum would pay a further milestone of $25m in cash, and the drug could be launched before the end of 2014.
To Read the Entire Report Please Click on the pdf File Below