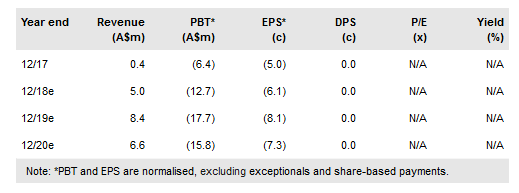
Telix Pharmaceuticals Ltd (AX:TLX) has assembled a portfolio of promising molecularly targeted radiation (MTR) therapeutic and imaging products for three different cancers. Each product has been validated by clinical studies or compassionate use in patients, thus reducing development risk. Telix is positioned to add significant value, such as by refining the products or developing them as a combination therapy. Preparations for a confirmatory Phase III study for kidney cancer imaging agent TLX250-CDx and for multiple Phase I/II studies of other agents are underway. It has begun commercialisation of an investigational prostate cancer imaging kit in the US, and plans are being developed for a short pivotal study to allow full approval. Our initial valuation is A$303m or A$1.39 per share.
Assembled a portfolio of advanced products
Telix’s MTR products comprise a radioactive isotope attached to either an antibody or small molecule that targets delivery to tumour cells in kidney, prostate or brain cancers (TLX250, TLX591 and TLX101, respectively). The companion diagnostic TLX250-CDx previously demonstrated high sensitivity and specificity for imaging clear cell renal cell carcinoma, the most common and aggressive form of kidney cancer, in a Phase III study. The product has been enhanced and the confirmatory ZIRCON Phase III will begin in Q418 on the completion of a short bridging study.
To read the entire report Please click on the pdf File Below: