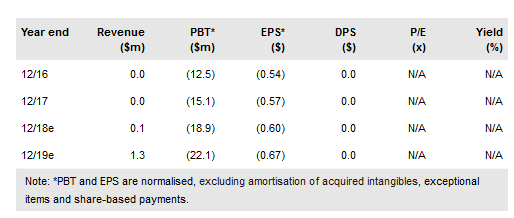
Volitionrx Ltd (NYSE:VNRX) recently announced the initial results from a 680-sample study taken from asymptomatic colorectal cancer (CRC) patients. The study demonstrated 80% sensitivity against stage 1 CRC and 66% against high-risk adenomas (at 78% specificity). The company has stated that it is moving on to the 4,300-sample study to determine the final test panel for European launch (results in Q218). This will be followed by a 12,000+ sample blinded study which is expected to complete in H218.
Data similar to previous results
The reported sensitivity and specificity for adenomas and stage 1 CRC reported in this study are similar to previous reports, in both symptomatic and asymptomatic patients. The company did not release data on detecting CRC as a whole. The panel in the study outperformed Epi proColon (62% sensitivity/81% specificity), but is difficult to compare to the widely used fecal immunochemical test (FIT), because FIT has substantially higher specificity (65% sensitivity/97% specificity).
To read the entire report Please click on the pdf File Below: