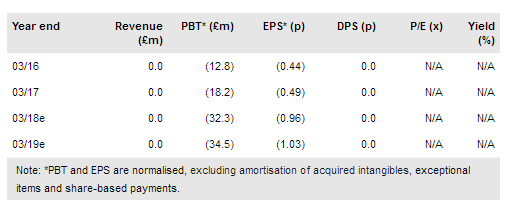
The highlight of FY17 was positive Phase II efficacy data from ReNeuron’s PISCES II clinical trial. As this and its other clinical programmes in ophthalmology and its earlier stage exosome platform progress, ReNeuron is entering a period of potentially significant milestones over the next 12 to 18 months. We maintain our valuation of £291m, adjusting for FY17 cash and rolling the model forward, which is offset by moving out the clinical timeline for the Phase III pivotal trial in stroke, now expected to start in early 2018 (vs H217).
Pivotal Phase III clinical trial planned early CY18
Reneuron Group (LON:RQE) received positive feedback from its end-of-Phase II meeting with the FDA for its Phase III stroke trial design. Alongside this it received a recommendation from the FDA to apply for a special protocol assessment (SPA) for the Phase III study. This is particularly encouraging as it indicates that, provided the endpoints are met, FDA approval should follow. ReNeuron has indicated that it now plans to apply for an SPA alongside a Regenerative Medicine Advanced Therapy (RMAT) designation (which would enable more interaction with the FDA during development and the possibility of accelerated review and approval) and an IND submission by the end of CY17 with the intention of starting a Phase III trial in the US in early 2018.
To read the entire report Please click on the pdf File Below: