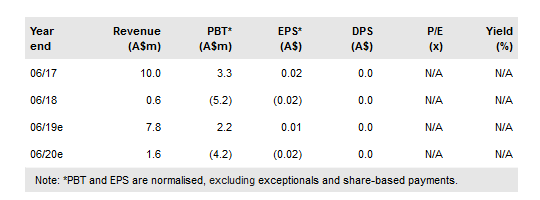
Regeneus Ltd (AX:RGS) has revised expected timing of an osteoarthritis (OA) clinical development licence deal for Progenza in Japan to Q4 CY18 (vs Q218); it is currently in active discussions with several parties. A licence deal would trigger a US$5m milestone payment from partner AGC and would effectively see Progenza commence a Phase II trial in knee OA in Japan. Regeneus is in separate discussions with potential licensees for other indications and territories for Progenza, as well as for its other pipeline products. We roll forward our rNPV model to FY19, which lifts our valuation to A$181m (vs A$170m) or A$0.87/share. Depending on the terms, we estimate an OA licence deal could add up to ~A$50m to our valuation.
Protracted negotiations preceded AGC deal in Japan
Regeneus is no stranger to protracted negotiations with Japanese companies. It successfully concluded a manufacturing licence agreement and JV with AGC in Japan in December 2016, after commencing partnering discussions the previous year, making it only the 17th Australian company to secure a significant technology licence in Japan in the past 20 years. It is well placed to achieve an OA licence deal in Japan, with a granted patent and manufacturing partner (AGC) in place, and promising signs of efficacy in the successful Phase I trial of Progenza in knee OA.
To read the entire report Please click on the pdf File Below: