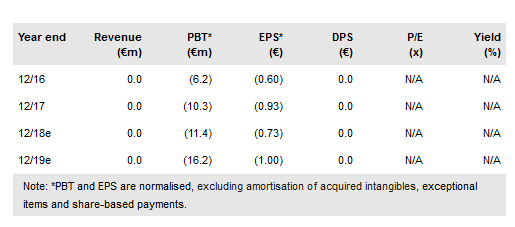
In April, Quantum Genomics SA (PA:ALQGC) announced it would be moving forward with the Phase IIb trial in heart failure and has recently announced the design of this study. The QUORUM study will assess the safety and efficacy of Quantum’s drug firibastat (QGC001) compared to ramipril (brand name Altace), an angiotensin-converting-enzyme (ACE) inhibitor, in 300 subjects enrolled within 24 hours after suffering acute myocardial infarction (AMI). The study is expected to launch by the end of 2018 with results due in H220.
Phase IIb heart failure trial design
The study will assess the safety and efficacy of firibastat compared to ramipril, an ACE inhibitor, in 300 subjects enrolled from 40 centres in the US and Europe within 24 hours of suffering AMI, also known as a heart attack. The primary endpoint will be the change from baseline in left ventricular ejection fraction (LVEF) after a three-month treatment. Secondary endpoints will include cardiac events, functional status and change in heart failure biomarkers.
To read the entire report Please click on the pdf File Below: