Probiodrug's (AS:PDB) new preclinical data showed that PQ912 demonstrated efficacy in Huntington’s disease (HD) in an animal model. Subject to further preclinical work, PQ912 could be fast-tracked to the clinic, which would diversify Probiodrug’s R&D pipeline with a new indication. Much will depend on the outcome of the company’s milestone clinical Phase IIa SAPHIR trial with PQ912 for Alzheimer’s disease (AD) with the data readout shortly. PQ912 is a first-in-class small molecule glutaminyl cyclase (QC) inhibitor and represents a differentiated approach in the AD field. We value Probiodrug at €345m or €42.1/share.
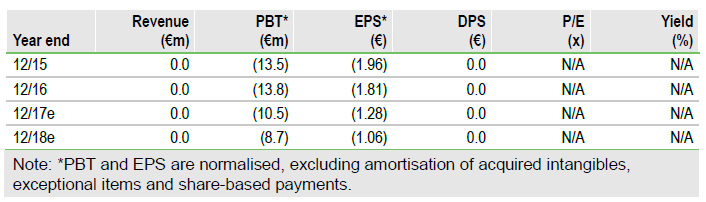
FY16 results in line with expectations
Probiodrug reported 2016 results in line with our estimates. R&D spend in FY16 was €11.0m, vs our expectation of €11.1m, while G&A expenditure of €2.9m was lower than our €3.3m estimate indicating good cost management. Probiodrug reported cash of €21.9m at end FY16, which included a capital raise of €14.9m in October 2016. The company guided that the net loss in 2017 could be lower than in 2016 (€13.9m) since R&D costs will decrease with the SAPHIR trial winding down.
To read the entire report Please click on the pdf File Below