EMA details clinical path for IMP321 in MBC
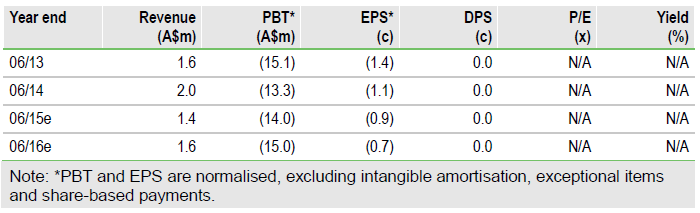
Prima Biomed Ltd (NASDAQ:PBMD) has received endorsement from the European regulator (EMA) for its planned Phase IIb trial of IMP321 in metastatic breast cancer (MBC). The primary endpoint is progression free survival. Our valuation and financial forecasts are under review pending funding resolution at the upcoming EGM and completion of the ongoing share purchase plan (SPP).
Phase IIb IMP321 study should start this year
The EMA has indicated that it considers the planned Phase IIb study (AIPAC) for Prima’s lead compound IMP321 (a soluble LAG-3 fusion protein) to be well designed, and if it achieves certain clinical end-points, sufficient to support a marketing authorisation. The study targets c 200 patients with HER-2 negative MBC planned over three years, starting with a safety run-in study in Q415. In a Phase I/IIa post-chemo study of IMP321, the six-month tumour response rate was 50% vs 25% in a historical chemo-only group. Prima also plans to launch a Phase I study of IMP321 in combination with an immune checkpoint inhibitor in Q415.
CVac data support search for a suitor
Meanwhile, final data from Prima’s Phase II CAN-003 study of dentritic cell vaccine CVac in second remission ovarian cancer reported in May, imply a minimum 16- month median overall survival (OS) advantage in patients treated with CVac vs standard of care. Median OS was not reached in the CVac group at 42 months vs 25.53 months in the standard treatment group. Partners are being sought.
To Read the Entire Report Please Click on the pdf File Below