Q116 results and the corporate update on 12 May showed that Oryzon Genomics SA (MC:ORY) is on track with its both clinical stage assets. Preliminary efficacy data from Part 2 Phase I/IIa study with ORY-1001 for acute leukaemia is expected to be announced at ASH in December 2016. Oryzon’s second product, ORY-2001, targets Alzheimer’s disease (AD) with preclinical data potentially indicating a disease-modifying effect. A Phase I trial started in Q116 and we expect data in early 2017. Our valuation is largely unchanged at €160m.
New debt boosts cash position
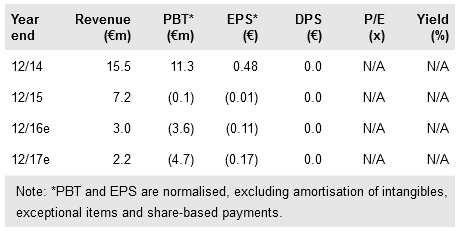
Oryzon reported its Q116 results on 12 May. Operating costs amounted to €1.8m (of which R&D expenses were €1.2m), which largely met our expectations. The cash position was significantly boosted (by €10.5m in new debt) to €29.0m and the additional funds will mainly be used to support existing operations. Oryzon has a history of efficient use of available public grants, which could further provide non-dilutive financing. In total, during the past 12 months Oryzon has managed to attract €27m in new funding from various sources.
To read the entire report Please click on the pdf File Below