Oryzon Genomics SA (MC:ORY) has two assets, ORY-1001 and ORY-2001, in the clinical stage of development, with ORY-3001 being investigated for oncology indications in the preclinical stage. While the most advanced, ORY-1001, is now in Roche’s hands, Oryzon has been making progress with the newer programmes. Data from the Phase I trial with ORY-2001 for Alzheimer’s disease (AD) will be announced by end March 2017 and fresh preclinical data from ORY-2001 studies support its potential in a second indication, multiple sclerosis (MS). We make minor adjustments to our valuation, increasing it slightly to €266m.
Financials: FY16 EBIT matches our estimate
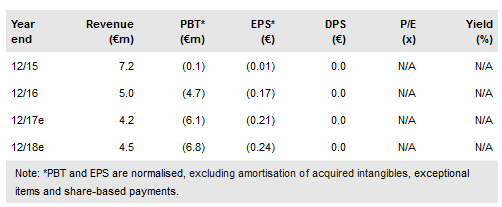
Oryzon’s FY16 results released on 24 February 2016 showed full year revenues at €735k (our estimate was €915k), consisting of a reimbursement payment from Roche and the recognition of deferred income. In addition, the company recorded €4.3m in income to account for the capitalisation of development costs, higher than our expected €3.9m. FY16 R&D costs of €5.2m were slightly ahead of our estimate of €4.8m, while personnel expenses of €2.5m were spot on. Adding other operating costs, Oryzon’s operating loss was €4.6m, close to our expectation of €4.7m.
To read the entire report Please click on the pdf File Below