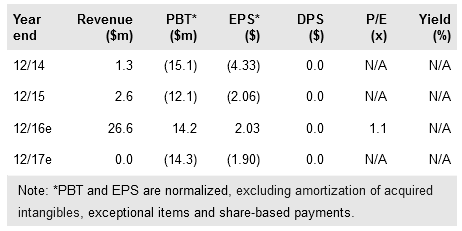
On 2 March Opexa Therapeutics (NASDAQ:OPXA) announced it has embarked on a restructuring initiative to prioritize its spending towards completing the ongoing 190-patient Abili-T Phase II study of lead candidate Tcelna in secondary progressive multiple sclerosis (SPMS). The company has reduced its overall headcount by about 30%, but this does not affect the Abili-T study timelines, for which data are expected in early Q416. The restructuring lengthens the company’s financial runway into Q117 (vs prior guidance of Q416), providing added flexibility around the timing of Tcelna data. Our rNPV is $35.1m, which after including $12.4m Q415 net cash equates to an equity valuation of $6.81 per share.
Abili-T final dose in February, data planned in Q416
The final patient dose of Tcelna in the Abili-T study was administered in February 2016, and approximately 97% of expected patient visits have been completed to date. Patients receive two annual courses of Tcelna treatment consisting of five subcutaneous injections per year at weeks zero, four, eight, 12 and 24. The primary endpoint is the percentage of brain volume change (whole brain atrophy) at 24 months. The firm has guided that it plans to release top-line data in early Q416; if data are positive, we anticipate Merck (NYSE:MRK) will exercise its option to license into the program, potentially triggering a $25m milestone payment to Opexa.
To read the entire report Please click on the pdf File Below