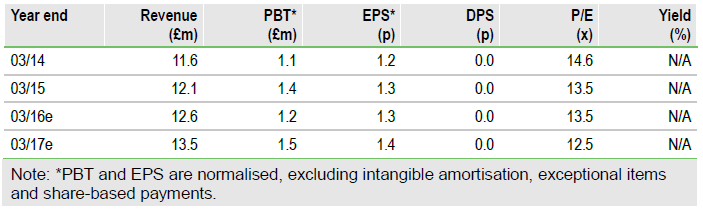
Allersys launch targeted for spring 2016
Omega Diagnostics Group Plc (L:ODX): H1 results (30 November 2015) noted that the Allersys allergy system could launch in spring 2016, with 37 optimised assays out of 40 targeted for launch. Omega’s Visitect CD4 test to monitor HIV patients shows a variability related to the ambient temperature. The cause is now known and a solution is being developed; the launch date is still uncertain. An Indian production facility in Pune starts operation in FY17 and should lead to sales gains in infectious disease products. Overall H1 sales were up 8% at £6.2m. Adjusted EPS was higher at 0.6p vs 0.5p due to a £135k tax credit.
Allersys: On track for spring 2016
The Allersys range runs on the Immunodiagnostic Systems (IDS) iSYS system using modern chemistry and magnetic particles. An initial customer evaluation in Spain on 10 allergens with 400 samples showed a linear relationship with ImmunoCap. Italian, German and French studies are planned, some with IDS. There are now 37 optimised allergens with 26 fully verified; 40 are being targeted for launch. IDS’s partners have developed autoimmune tests that run on iSYS and complement allergy tests. Management indicates a spring 2016 launch.
To Read the Entire Report Please Click on the pdf File Below