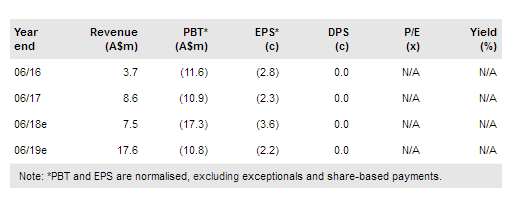
Novogen Ltd (NASDAQ:NVGN) is on track to commence a Phase II trial of GDC-0084 in glioblastoma in Q4 CY17, in line with previous guidance. It plans to meet with the FDA to discuss trial design as it finalises preparations for the study. A Phase I trial of Cantrixil in ovarian cancer is well underway, with initial data expected in late 2017 or early 2018, efficacy data from an expansion cohort at the MTD are likely in H2 CY18. We lift our valuation range slightly to between A$87m and A$152m (A$0.18-0.31 per share), while noting that additional funds may be needed in FY18.
GDC-0084 Phase II to start in Q417
Novogen is on track to initiate a Phase II trial of GDC-0084 (a small molecule inhibitor of the PI3K/Akt/mTOR pathway) in glioblastoma (brain cancer) in Q417. It has appointed Chiltern Oncology to manage the trial, and the manufacture of finished dosage capsules of the drug is almost complete. Consultations with the FDA to discuss key features of the clinical trial design are expected before the study commences. The trial will test GDC-0084 in a subset of glioblastoma patients who are known to obtain little benefit from standard temozolomide chemotherapy; the study is supported by encouraging Phase I data. The clear unmet medical need in this patient group could open access to accelerated approval pathways.
To read the entire report please click on the pdf file below: