NovaBay (NBY) has raised $7m in a public offering of common stock and one-year warrants in advance of a pivotal year – 2013 will see three Phase II trial read-outs for its topical anti-infective, NVC-422, offering re-rating potential and further financing/partnering opportunities. NovaBay’s estimated FY12 cash reserve of $17m now extends to mid-2014 and enables accelerated enrolment in the ongoing viral conjunctivitis trial and new pipeline developments such as the start of a Phase IIa study in bacterial conjunctivitis. NVC-422 could therefore become a one-product treatment for both viral and bacterial conjunctivitis, a significant commercial opportunity.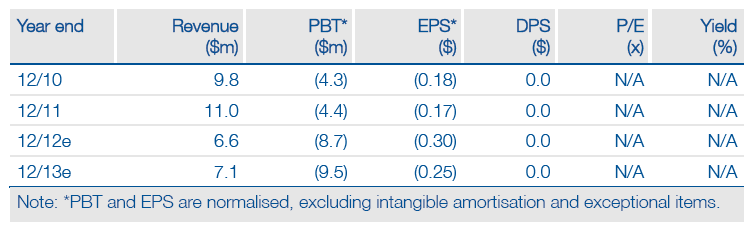
$7m cash injection, with possible $6.6m from warrants
NovaBay sold 5.9m shares of common stock through a public offering at $1.25 per share (10% discount), as well as one-year warrants to purchase up to 4.425m shares (strike price $1.50/share). Net proceeds from the offering was $7m, while a further $6.6m could be received if the newly issued warrants are fully exercised.
Catalysts in sight
NVC-422, a so-called Aganocide compound with disruptive potential, is being studied in three Phase II studies that should read out in 2013. Data from a 450-patient trial in viral conjunctivitis in H213 is a major potential share price and partnering catalyst, while positive results from a 300-patient impetigo study in mid-13 would trigger a further single-digit, $m milestone (c $3.5m) from partner Galderma. Data from 20-40 patients in the proof of concept Phase IIa study for urinary catheter blockage and encrustation should be available in H113. Global partners are also being sought for NeutroPhase, a wound-cleansing agent with FDA 510(k) clearances – Pioneer Pharma is a strategic partner (5.6% shareholder) in China/South-East Asia.
Valuation: Increased to $66m from $60m
We have increased our sum-of-the-parts DCF valuation of NovaBay to $66m (previously $60m) to include the equity offering, although the per share value drops to $1.79 (vs $1.96) based on 36.9m shares now outstanding. This represents upside to NovaBay’s market capitalisation of $42m and $1.13 share price. We also note further upside potential from successful development of NVC-422 for bacterial conjunctivitis, a ~$1bn global market opportunity which we do not currently include in our valuation model. FY12 net cash is estimated at $17m and should reach to mid-2014, enabling key clinical milestones in 2013 to potentially re-rate the stock. Further injections of cash could be receivable from issued warrants and development milestones/options exercised by existing partners (Galderma and Pioneer Pharma).
To Read the Entire Report Please Click on the pdf File Below.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
NovaBay Pharmaceuticals Offering Should Help Support Pivotal Year
Published 12/20/2012, 02:16 AM
Updated 07/09/2023, 06:31 AM
NovaBay Pharmaceuticals Offering Should Help Support Pivotal Year
Bases loaded for a pivotal year
3rd party Ad. Not an offer or recommendation by Investing.com. See disclosure here or
remove ads
.
Latest comments
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.