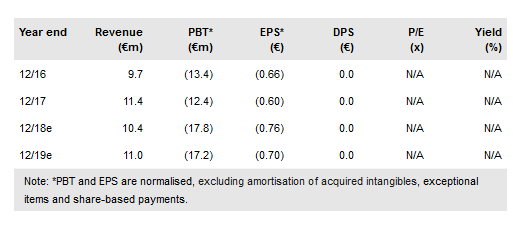
In H118, Medigene NA O.N. (DE:MDG1k) announced several key developments including the start of its MDG1011 Phase I/II TCR clinical trial, a gross capital raise of €32.3m (which expands cash reach past 2019) and the expansion of its bluebird partnership to cover a total of six targets (worth potentially US$1.5bn in milestones plus royalties). The MDG1011 Phase I/II clinical trial is enrolling patients and we forecast that the Phase I part of the TCR trial and the now fully enrolled Phase I/II DC vaccine trial will read out in 2019. We have updated our forecasts and now value Medigene at €453m (€18.47/share).
TCR trial: Aiming to increase enrolment speed
Medigene has begun enrolment for its first clinical trial (Phase I/II) with its T-cell receptor (TCR) modified T-cell (MDG1011) product candidate in patients with r/r multiple myeloma (MM), acute myeloid leukaemia (AML) or myelodysplastic syndrome (MDS). Medigene has noted that it is in discussion with the German regulatory authority with the aim of speeding up enrolment by changing the inclusion criteria from one patient per disease in each dose cohort to require at least one MM patient and at least one patient of either AML or MDS.
To read the entire report Please click on the pdf File Below: