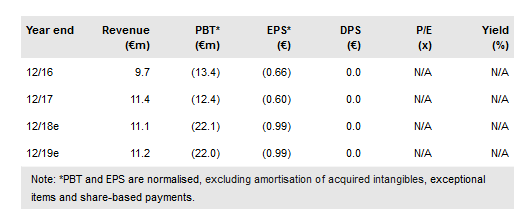
Medigene NA O.N. (DE:MDG1k) has announced an expansion of its bluebird partnership, giving significant external validation of its T-cell receptor (TCR) technology platform. The full partnership now covers six targets and is worth potentially US$1.5bn in development and commercial milestones, in addition to royalties on any future sales. Medigene’s own internal pipeline continues to advance with its MDG1011 Phase I/II TCR clinical trial now enrolling patients. Additionally, we forecast that the Phase I part of the TCR trial and the now fully enrolled Phase I/II DC vaccine trial will read out in 2019. We have updated our forecasts and now value Medigene at €416m (€18.65/share) vs €396m (€17.8/share) previously.
bluebird expansion further validates technology
In September 2016, bluebird and Medigene signed a partnership agreement to develop four TCR product candidates. This deal has now been expanded to include an additional two TCRs, taking the total to six ongoing programmes. The expansion includes a one-off payment of €8m to Medigene and up to €250m in potential milestone payments per candidate. Medigene has a unique platform for identifying and characterising specific TCRs to target antigens. The selection of the correct antigen/major histocompatibility complex (MHC) restriction combination is a complex undertaking and we believe this expanded agreement further validates Medigene’s expertise in this area.
To read the entire report Please click on the pdf File Below: