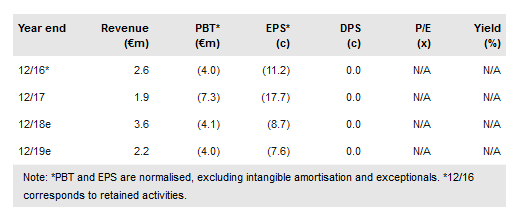
Hybrigenics (PA:ALHYG) has published data from a Phase II study in chronic myeloid leukaemia (CML). After one year of treatment, 40% of patients showed improvements beyond a major molecular response (MMR) and 20% achieved a deep molecular response (DMR), consistent with a functional cure. The ongoing Phase II trial in acute myeloid leukaemia (AML) will complete enrolment before end 2018, with data readout in 2019. Hybrigenics has completed its refocusing on R&D with the sale of the genomics division, Helixio. The internal R&D programme on ubiquitin-specific proteases (USP) inhibition continues. Hybrigenics also has an R&D partnership with Servier focused on oncology worth up to €12m. Cash at end 2017 was €7m. Our valuation is €129.5m or €2.77/share.
CML trial reads out; AML data on track for 2019
In CML a DMR may be considered a functional cure and stopping treatment (even temporarily) can be an option. The Phase II study in CML showed that 40% (8/20) of patients had a decrease of BCL-ABL beyond 3 log, which is an MMR. A decrease of more than 4.5 log was achieved by 20% (4/20) of patients. These data are lower than previous interim data released in February 2017 where MMR was 43% and DMR was 33%. Separately, the Phase II study in AML in combination with decitabine has enrolled 87 of the planned 110 patients. The company expects to complete enrolment by this year’s end and report results in 2019. To maximise the cash runway, Hybrigenics will assess the next studies in CML, chronic lymphocytic leukaemia (CLL) and prostate cancer once the AML data are announced.
To read the entire report Please click on the pdf File Below: