Hybrigenics’ (ALHYG.PA) main value driver remains inecalcitol, based on its potential in oncology, while its development in its former key indication, psoriasis, is on hold. Key data from a Phase II trial in chronic lymphocytic leukaemia (CLL) are due in 2013 and could lead to a licensing deal, and potentially, resumption of trials in castration resistant prostate cancer (CRPC). Meanwhile, Hybrigenics is expanding its service division to reduce its dependence on inecalcitol and recently launched its novel small molecule screening service, YChemH. Our base case valuation is reduced to €23m excluding psoriasis, rising to €29m if trials in this indication resume.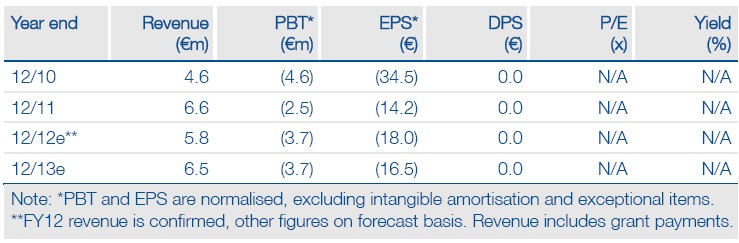
Important data on inecalcitol expected in 2013
Interim data from the Phase II proof-of-concept trial with inecalcitol in CLL are due in H113 and final data could be reported in Q413. If the results suggest that the compound delays progression of the disease, this should lead to the partnering of the product and the resumption of its development in CRPC. The strategy for inecalcitol in psoriasis should also become clearer once the CLL results are reported.
Expanding the services division
Hybrigenics has initiated its expansion strategy for its services division with the launch of YChemH, a novel small molecule screening service. The new service was developed with UK medicinal chemistry company, Charnwood Molecular and is the first step towards creating a more integrated range of services in order to reduce its dependence on inecalcitol. Further expansion either organically or via M&A is likely.
Financials, headline sales figures
Total FY12 revenue was €5.8m, including a €0.96m grant. Hybrigenics received a €1.7m upfront payment from Servier in 2011, so total revenue fell 12% from €6.6m. Headline recurring revenue (excluding the grant) stood at €4.8m, flat on 2011. Services revenue fell c 2% at €3.3m, (H1 sales fell 16% and recovered c 9% in H2).
Valuation: Base case DCF of €23m
Our base case valuation is €23m, excluding inecalcitol in psoriasis as it is now on hold (previously €51m, included €31m for the psoriasis indication). We now value the psoriasis programme at only €6m after reviewing our assumptions. We value the services division on 2x FY13e sales at €7.2m. The company had cash of €3.5m at end-December 2012 and a further €6.9m is available from the Yorkville equity line, which could finance the company through to late 2014.
To Read the Entire Report Please Click on the pdf File Below.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
Hybrigenics: Main Value Driver Remains Inecalcitol
Published 02/11/2013, 12:24 AM
Updated 07/09/2023, 06:31 AM
Hybrigenics: Main Value Driver Remains Inecalcitol
Hybrid business model
3rd party Ad. Not an offer or recommendation by Investing.com. See disclosure here or
remove ads
.
Latest comments
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.