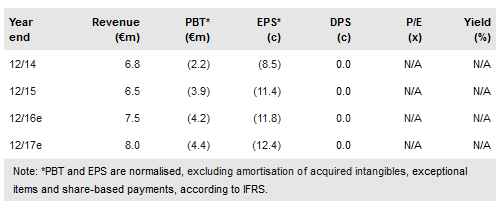
Hybrigenics (PA:ALHYG) has presented the first ever clinical data from an interim analysis of a Phase II study of inecalcitol in combination with imatinib in patients with chronic myeloid leukaemia (CML). At interim, 43% of patients who had been on treatment for three months showed improvements to baseline MMR (major molecular response). Furthermore, 33% of patients who had completed one year in the study achieved a deep molecular response (DMR). Achieving and sustaining a DMR may allow patients to discontinue treatment (functional cure). Therefore, we believe these are encouraging preliminary data that provide first clinical insights into inecalcitol’s potential in CML. The study is expected to complete in H218. Our valuation stands at €146.1m.
Improvements in 43% of patients
The efficacy endpoint in this Phase II study is the proportion of responders, defined as patients achieving DMR within 12 months of inecalcitol treatment. Achieving and sustaining DMR may be considered a functional cure and stopping treatment can be an option. Other studies report that 1-3% of imatinib-treated patients achieve DMR after 12 months of treatment, increasing to 31-33% after five years. Thus, 33% of patients in this trial achieving a DMR with the addition of inecalcitol within 12-months is encouraging.
To read the entire report Please click on the pdf File Below