GW Pharmaceuticals’ (GW) stock is trading at a 52-week low despite having achieved key commercial and R&D milestones in 2012, meeting financial expectations, while maintaining a solid financial base. On our valuation, the current share price discounts potential new indications for Sativex (cancer pain), the entire R&D pipeline and possible licensing deals (Otsuka). As such, we see a buying opportunity ahead of potential value-inflection points, both commercial and clinical, in 2013.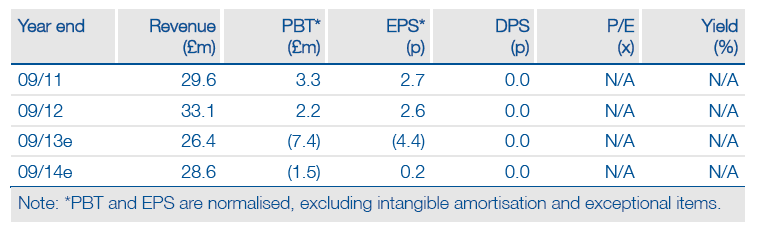
Sativex Commercial: Steady Progress In 2012
Encouraging in-market sales growth (£10.0m in FY12, +89%) reflects continued uptake in existing markets and new country launches. In 2013, we expect the completion of German pricing negotiations (Q113), an Italian launch (Q113) and additional EU roll-out (mid-2013 onwards) to create a strong platform for future growth. Beyond Europe, Sativex sales growth could be supported by launches in Australia (Q413/Q114) and Middle-Eastern territories (Q413 onwards).
R&D: Expect A Busy 2013
GW has announced encouraging Phase II results for GW42004 (novel oral cannabinoid) in Type II diabetes. Initial signs of efficacy and acceptable safety pave the way for a larger Phase IIb dose-ranging study in H213. In 2013, we also expect three programmes to enter clinical trials (for schizophrenia, epilepsy and glioma), further Phase II data (ulcerative colitis) and, potentially, headline Sativex Phase III results (cancer pain) in Q413. Finally, completion of the Otsuka cancer/CNS collaboration in mid-2013 could be an important inflection point for licensing the lead candidates.
Financials: Fine-Tuning Sativex Forecasts
We have updated our FY13 numbers and published new FY14 forecasts following GW’s guidance and revised timelines for further EU launches of Sativex. The main changes to our model are lower Sativex sales for FY13-15 reflecting the protracted European pricing negotiations and higher R&D spend. Despite a more gradual EU roll-out, our view of Sativex peak sales potential (£64m/year) remains unchanged.
Valuation: DCF-Based Valuation Of £195m
We value GW at £195m, or £1.46 per share, based on a DCF analysis. Sativex in MS spasticity contributes 53p per share, with cash adding another 13p. Thus, the current share price of 58p appears to discount any new Sativex indications, pipeline success, or potential licensing deals, which in our view represents a clear buying opportunity.
To Read the Entire Report Please Click on the pdf File Below.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
GW Pharmaceuticals: Multiple Value Inflection Points In 2013
Published 12/13/2012, 02:40 AM
Updated 07/09/2023, 06:31 AM
GW Pharmaceuticals: Multiple Value Inflection Points In 2013
Bang For Your Buck
3rd party Ad. Not an offer or recommendation by Investing.com. See disclosure here or
remove ads
.
Latest comments
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.