Yesterday the U.S. Commerce Department released the widely-anticipated first quarter GDP data.
As expected, the U.S. economy contracted in the first quarter as GDP fell 4.8%. It was the first negative GDP print since the first quarter of 2014 when GDP fell 1.1%. The nearly 5% drop in GDP in the first quarter marks the worst GDP reading since the fourth quarter of 2008 when GDP fell 8.4%.
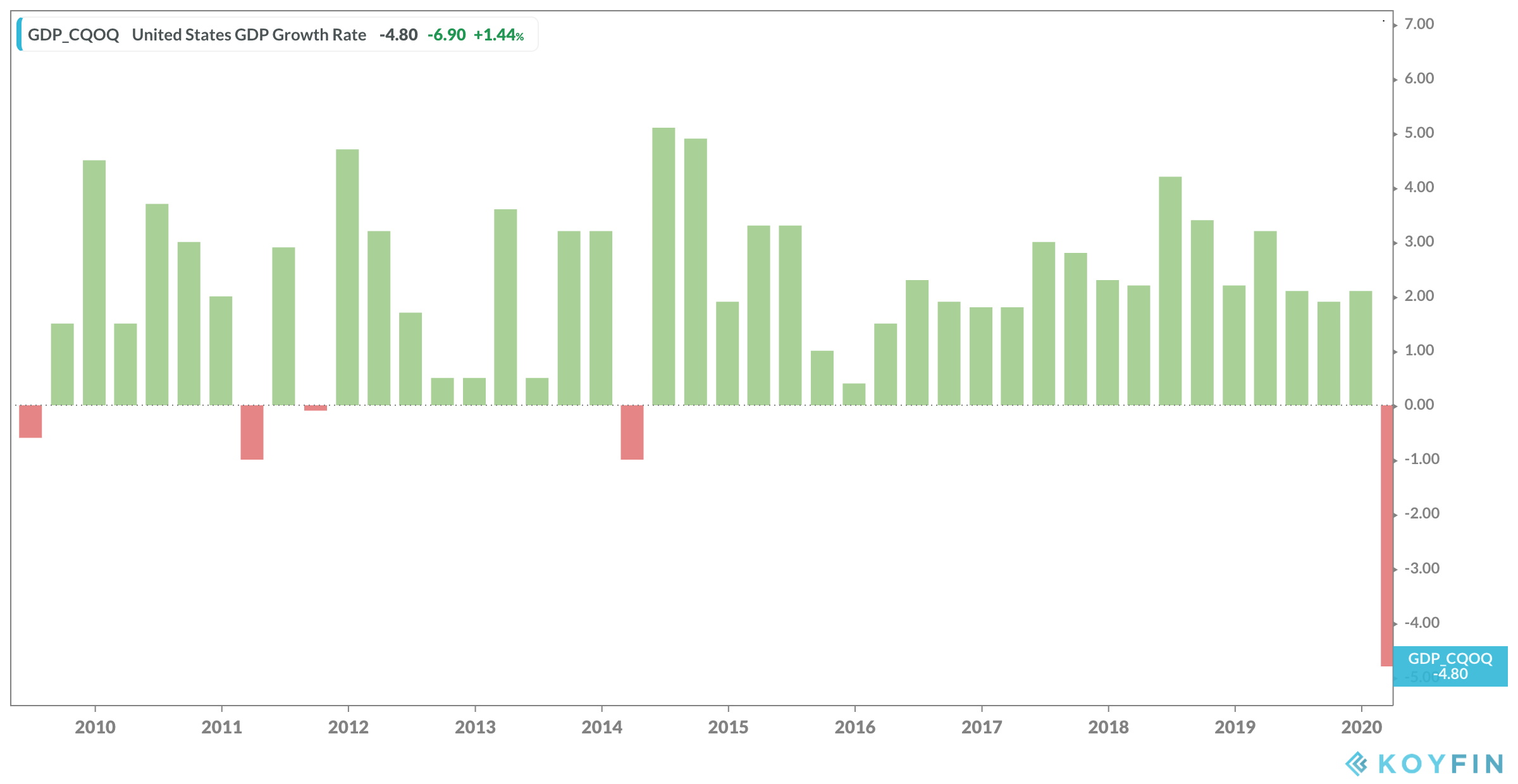
Consumer spending, which makes up more than two-thirds of U.S. economic activity, fell 7.6% in the first quarter. The drop in consumer spending marked the largest quarterly drop since the second quarter of 1980.
The Commerce Department's Bureau of Economic Analysis cited shelter-in-place orders as negatively affecting GDP for the quarter, saying they "led to rapid changes in demand, as businesses and schools switched to remote work or canceled operations, and consumers canceled, restricted, or redirected their spending."
As bad as all of that sounds, markets soared yesterday as headlines about Gilead Sciences' (NASDAQ:GILD) antiviral drug remdesivir conveniently came out just as the GDP data was released.
Gilead said preliminary results of the drug to treat COVID-19 showed at least 50% of patients who had been treated with a five-day dosage saw improvement. Gilead also said a separate trial being conducted by the National Institute of Allergy and Infectious Diseases met its main goal, although didn't provide further details.
"These data are encouraging as they indicate that patients who received a shorter, five-day course of remdesivir experienced similar clinical improvement as patients who received a 10-day treatment course," lead investigator of the study Aruna Subramanian said.
Scott Gottlieb, the former commissioner of the Food and Drug Administration, warned the drug is "not a home run, a cure by any means." Gottlieb continued,"It’s not going to be a cure, but it is going to be a drug potentially that if you use it particularly early in the course of the disease ... it could reduce their chances of having a really bad outcome."
With hopes for remdesivir high, the Food and Drug Administration is in talks with Gilead to make remdesivir available to COVID-19 patients.
"As part of the FDA’s commitment to expediting the development and availability of potential COVID-19 treatments, the agency has been engaged in sustained and ongoing discussions with Gilead Sciences regarding making remdesivir available to patients as quickly as possible, as appropriate," an FDA spokesperson said.
For the sake of humanity, hopefully remdesivir is as effective as the markets are interpreting it to be.
