Further VIPES responder analyses at EAACI
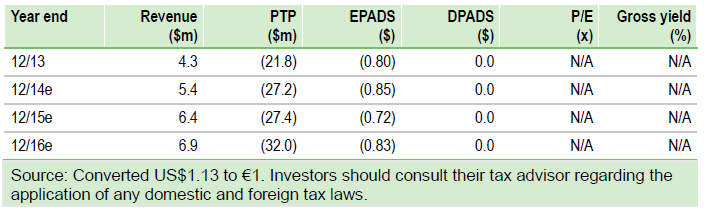
Additional data from DBV Technologies (PARIS:DBV)’s Phase IIb VIPES trial have been presented at EAACI. A further post hoc analysis applying more rigorous efficacy criteria reinforces the strength of the existing data, in our view. DBV also hosted a well-attended physician symposium, highlighting interest in DBV’s Viaskin products and helping to raise broader awareness, both key for a successful launch. Our valuation is largely unchanged at $1,357m.
Further VIPES analyses presented at EAACI
A new post hoc analysis of VIPES presented at EAACI applied a more rigorous definition of “responder” to the existing dataset. This latest analysis demonstrates that 50% of children (aged 6-11 years) at the highest Viaskin dose responded, compared to 12.9% on placebo (p=0.0039). These new data largely exclude highly sensitive children who may have only had a small increase in the actual quantity of peanut protein consumed, compared to baseline (eg from 1mg on entry to 10mg at 12 months, meeting the >10-fold increase responder definition). These data add to both previously published top-line data and detailed data made available at AAAAI.
To Read the Entire Report Please Click on the pdf File Below