Five things to watch in markets in the week ahead
Crossject (PA:ALCJ) has developed a deep pipeline of products that are based on its proprietary needle-free injection system, Zeneo, across a variety of indications, with a focus on emergency-related areas. In addition to avoiding needles, Zeneo provides a simple and quick (~1/10th of a second) delivery of the drug. Recently, the company has added Zeneo Terbutaline for the acute treatment of exacerbations in severe asthmatics. This is a large market (~2.5m in the US) in need of better choices for patients.
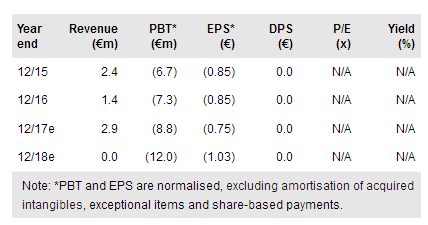
US partnership possible by year-end 2017
Crossject expects to license its lead product Zeneo Sumatriptan for the acute treatment of migraine in the US by YE17. Ease of use and fast onset should be key selling points for the product. Additionally, a tender process for US rights for Zeneo Midazolam for the acute treatment of epilepsy is expected to begin by year-end.
To read the entire report Please click on the pdf File Below: