Clavis Pharma (CLAVIS.OMX) :The CLAVELA study established that elacytarabine has no benefit over cytarabine in the management of third-line relapsed and refractory acute myeloid leukaemia. The hypothesis was that by attaching a lipid molecule to cytarabine, elacytarabine would have improved pharmacokinetics giving a better outcome. Since the other lead, a lipid gemcitabine (CP-4126) failed last November, Clavis is now expected to enter an orderly wind up. There is not expected to be much cash for shareholders. An alternative reversal into a private Scandinavian biotech will be investigated.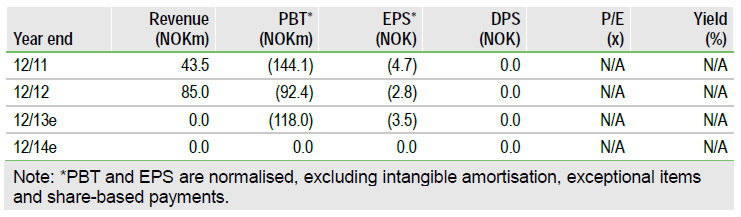
CLAVELA data
Median overall survival was 3.5 months vs 3.3 months in the seven possible alternative treatments, some of were palliative. There were no obvious subgroups showing better efficacy. The response rate was 23% vs 21%. The hazard ratio was 0.97, p=0.73. In the 60 patient Phase II, a 5.3 month median overall survival was seen vs 1.5 months in historic controls with an 18% response rate.
Why didn’t it work?
One can speculate, that whilst attaching lipids to proven anti-cancer stalwarts like gemcitabine (in LEAP) and cytarabine (in CLAVELA) certainly improves pharmacokinetics and maybe overcomes some drug transporter resistance mechanisms, the fact that the metabolic clearance pathways were unaffected once the lipid moiety was cleaved (necessary for activity), offset the advantages. This is particularly the case in third-line patients who had previously failed on cytarabine and so already had some level of resistance. The doses used were clinically equivalent. In CLAVELA, longer exposure times may have been offset by efficient metabolic deactivation. The failures show that small cancer Phase II studies against historic controls are unreliable Phase III guides. It also shows that pitting a single agent against combination therapy in terminal disease is very risky.
Valuation: Little cash or a private reversal?
At the embers of the pyre cool, shareholders will find a meagre hoard of gold for distribution. Net cash after payment of creditors is estimated to be NOK140m out of NOK223m cash as at 31 December. There will also be the final CLAVELA costs to settle and the redundancies to fund plus other close down costs. Edison has a speculative forecast of approximately NOK30m residual cash for distribution, about NOK1 per share; this might be pessimistic. An alternative idea of a reversal into Clavis by a private Scandinavian biotech company has been mooted. The main value in Clavis would be some cash plus the current listing and a clinical development team in cancer.
To Read the Entire Report Please Click on the pdf File Below.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
Clavis Pharma:Little Cash Or A Private Reversal?
Published 04/04/2013, 07:51 AM
Updated 07/09/2023, 06:31 AM
Clavis Pharma:Little Cash Or A Private Reversal?
Latest comments
Loading next article…
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.