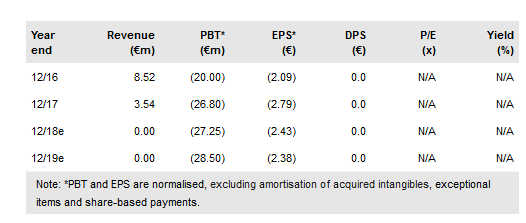
Celyad SA (NASDAQ:CYAD) has presented an update on its CYAD-01 solid cancer trials with haematological data due in early December. There are strong indications that optimal effects can be gained by use of high doses, possibly with Cy-Flu preconditioning, or by combining CYAD-01 with chemotherapy in solid cancer. An allogenic colorectal trial will start in early 2019; an allogeneic product (CYAD-101) could have a major market in multiple cancer types. Our indicative value remains at €1,090m (€89/share) pending more data.
Leading position in solid cancers and allogeneic cells
NKR CAR T-cell therapy targets “stressed” cancer cells. In early data (at SITC in November) CYAD-01 with prior FOLFOX (which stresses cells) in metastatic colorectal cancer (mCRC) gave one complete and two partial pathological responses at a 3x108 dose; the study continues. In stand-alone therapy, three patients who had peak CYAD-01 blood levels of over 7,000 cells/ml showed stable disease (SD). Initial data from a trial using Cy-Flu preconditioning indicates much higher peak dose cell levels; more data is due in mid-2019. By year-end 2018, the first mCRC patients will be enrolled in a trial with allogeneic cells (CYAD-101) following FOLFOX. Potentially, CYAD-101 offers a more affordable, rapid -response therapy for thousands of patients. Celyad holds key patents in allogeneic therapy (licensed by Novartis). A shRNA approach (licensed from Horizon Discovery) is in preclinical development. These developments give Celyad a leading position in solid cancers, an area other CAR therapies find hard to access (see 2017 report).
To read the entire report Please click on the pdf File Below..