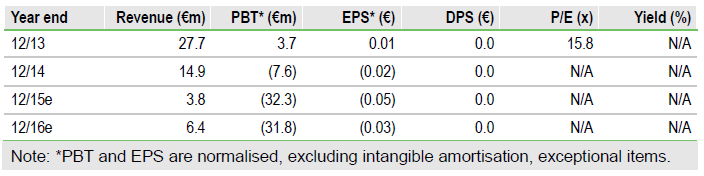
Pipeline progressing on track
Initial recruitment is on track for the Phase III pivotal study (TOZ-PD) of Biotie Therapies Oyj (O:BITI) lead product candidate, tozadenant. Tozadenant offers a novel mechanism of action for the treatment of Parkinson’s disease (PD) patients experiencing motor fluctuations on levodopa. Top-line data from the double-blind portion of the trial are expected by end 2017. Meanwhile, two other products, SYN120 (Parkinson’s disease dementia, PDD) and BTT1023 (primary sclerosing cholangitis, PSC), are in Phase II studies; data are expected by end 2016. We value Biotie at €273m, or €0.28/share.
Tozadenant in pivotal territory
TOZ-PD, along with the completed Phase IIb study, would form the basis of an NDA submission for tozadenant. Agreed under Special Protocol Assessment with the FDA, it is divided into two phases: a 24-week, placebo-controlled phase (n=450), followed by a 52-week, open-label phase. If the primary endpoint is met in the placebo-controlled phase, a second 52-week, open-label study will be performed (n=450) to generate the requisite 1,500 unique patient exposures for approval.
To Read the Entire Report Please Click on the pdf File Below