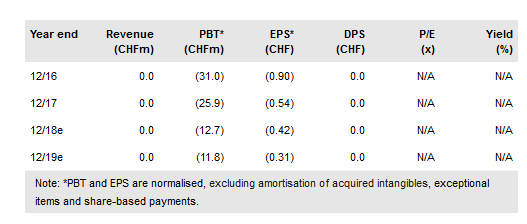
Auris Medical Holding AG (NASDAQ:EARS) recently reported its Q318 results and provided a brief business update on its active programs. R&D expenditure for the period was down roughly 60% over Q317, which reflects management’s swift pivot to focus on Phase I development. Auris plans to initiate two intranasal betahistine trials in Q119: the AM-125 Phase II trial in acute vertigo and AM-201 Phase I trial for olanzapine-induced weight gain.
Upcoming Phase II AM-125 trial
Auris is developing AM-125, an intranasal formulation of betahistine for the treatment of acute vertigo. As AM-125 bypasses the digestive tract where the oral compound is readily metabolised, the intranasal formulation has demonstrated superior bioavailability over oral betahistine. Auris expects to initiate the Phase II trial in Q119 in 138 patients with surgically induced acute vertigo following vestibular schwannoma excision.
To read the entire report Please click on the pdf File Below..