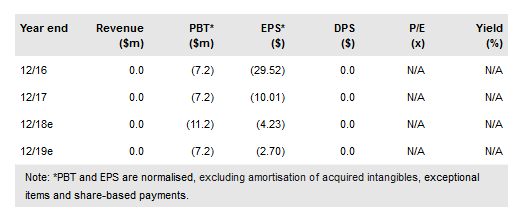
Atossa Genetics Inc (NASDAQ:ATOS) is preparing to start Phase II studies of both its oral and topical endoxifen formulations in Q218. Endoxifen, an estrogen receptor (ER) antagonist, is being advanced in topical form to treat high mammographic breast density (MBD), and also as an oral drug to prevent cancer recurrence in women refractory to tamoxifen. The company plans to raise $20m in equity through a rights offering in May 2018, which we believe should fund the Phase II endoxifen trials through completion.
More rapid steady-state levels with oral endoxifen
In February 2018, Atossa reported new data from its prior Phase I study, suggesting that the median time for patients to reach the steady-state serum levels of endoxifen while taking daily oral doses was seven days vs 50-200 days for patients taking oral tamoxifen daily. Oral endoxifen may thus provide therapeutic plasma endoxifen levels weeks or even months earlier than oral tamoxifen, which may potentially provide a more rapid onset of therapeutic effect.
To read the entire report Please click on the pdf File Below: