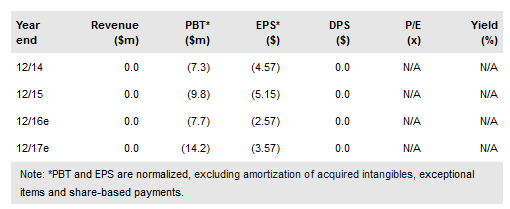
Atossa Genetics Inc (NASDAQ:ATOS) raised $2.875m in an equity raise in September 2016. We expect the proceeds to be allocated to Atossa’s two active pipeline programs: its 30-pt Phase II study on its intraductal microcatheter (IDMC) delivering fulvestrant to breast ducts in patients scheduled for mastectomy or lumpectomy, and its oral endoxifen candidate, expected to start human studies in 2017 as potential treatment for breast cancer patients refractory to tamoxifen. We obtain an rNPV-based equity valuation of $10.7m.
IDMC-fulvestrant interim data expected shortly
Atossa’s IDMC intends to deliver fulvestrant to the breast with potentially higher local exposure and lower systemic risks vs the established intramuscular delivery approach. We expect the firm to provide interim clinical data for the ongoing 30-patient Phase II study by Q117, after which we expect a larger pivotal study would be required for approval under the FDA 505(b)2 process. We assume the study would start in late 2017 or early 2018, with commercialization in 2020 and peak sales of $183m in 2025, and that Atossa would earn a 20% royalty on net sales.
To read the entire report Please click on the pdf File Below