AstraZeneca (NYSE:AZN) announced that the FDA approved the label expansion of its BTK inhibitor, Calquence (acalabrutinib), as a treatment for adult patients with chronic lymphocytic leukemia (“CLL”) or small lymphocytic lymphoma (“SLL”). The approval was based on positive data from two phase III studies — ELEVATE-TN and ASCEND — wherein Calquence demonstrated superior progression-free survival (PFS) in both first-line and relapsed or refractory CLL.
Calquence is presently marketed for the treatment of relapsed or refractory mantle cell lymphoma. The company has plans to submit regulatory applications seeking a label expansion of the same to include CLL patients in Europe and Japan in the first half of 2020.
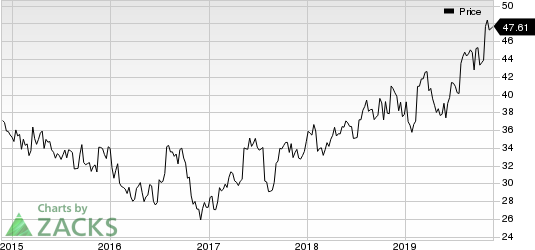
Shares of AstraZeneca have rallied 25.3% so far this year compared with the industry’s growth of 4.1%.

The ELEVATE-TN study evaluated Calquence monotherapy or in combination with Roche’s (OTC:RHHBY) Gazyva (obinutuzumab) in first-line CLL patients. Data from the study showed that treatment with the drug alone or in combination has reduced the risk of disease progression or death by 80% and 90%, respectively, compared to chemotherapy, chlorambucil, in combination with Gazyva (standard-of-care medicines). The median time to disease progression in patients has not been reached yet for both monotherapy and the combo regimen versus 22.6 months for standard-of-care.
The ASCEND study compared Calquence monotherapy to a combination regimen of Roche/Biogen’s (NASDAQ:BIIB) CD-20 antibody, Rituxan (rituximab), plus Gilead’s (NASDAQ:GILD) Zydelig (idelalisib) or Teva’s Treanda (bendamustine) in previously treated CLL patients. Previously announced data showed that Calquence reduced the risk of disease progression by 69% compared to the Rituxan combination therapy after a median follow-up of 16.1 months. The median PFS for the drug is yet to be reached, while the same for the Rituxan combo is 16.5 months. Meanwhile, 88% of patients treated with Calquence did not show any disease progression at 12 months compared to 68% for the control arm.
The safety and tolerability profile for Calquence was consistent with its established profile in both clinical studies.
Calquence generated $108 million in the first nine months of 2019. With the approval for this much larger indication, AstraZeneca can gain access to a broader patient population and boost sales of this promising drug.
AstraZeneca has built a strong oncology portfolio, which includes Tagrisso, Imfinzi, Lynparza, Iressa, Calquence and other legacy drugs. Sales of this segment were 37% of the company’s total sales in the first nine months of 2019. Oncology sales were up 50% year over year in that period.
Zacks Rank
AstraZeneca currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
The Hottest Tech Mega-Trend of All
Last year, it generated $24 billion in global revenues. By 2020, it's predicted to blast through the roof to $77.6 billion. Famed investor Mark Cuban says it will produce "the world's first trillionaires," but that should still leave plenty of money for regular investors who make the right trades early.
See Zacks' 3 Best Stocks to Play This Trend >>
Roche Holding (SIX:ROG) AG (RHHBY): Free Stock Analysis Report
AstraZeneca PLC (AZN): Free Stock Analysis Report
Biogen Inc. (BIIB): Free Stock Analysis Report
Gilead Sciences, Inc. (GILD): Free Stock Analysis Report
Original post
Zacks Investment Research