Promising data from Actimab-A Phase I/II study
Actinium (OTC:ATNM) is progressing two compounds, Iomab-B and Actimab-A, through the clinic for the treatment of blood cancers, with a further three programs in preclinical development. Actimab-A has successfully completed Cohort 3 of the first part of the Phase I/II trial underway in elderly patients with newly diagnosed AML. The encouraging clinical outcomes, coupled with a promising toxicity profile, suggest it should progress to Phase II once the maximum dose is established.
Actimab-A shows encouraging safety and efficacy
Actinium Pharmaceuticals has successfully completed the third patient cohort of Actimab-A’s Phase I/II study. The study is evaluating elderly patients (older than 60) with newly-diagnosed Acute Myeloid Leukaemia (AML). The aim of the first part of the study (n=c 24) is to determine the maximum tolerated dose (MTD), with low-dose cytarabine, for use in the second stage (n=53), which will assess efficacy. The third cohort involved an additional three patients, of which two achieved complete remission with differing degrees of haematological recovery (CRi). The result is impressive, especially in the context of such a high disease burden and the advanced age (up to 87) in the patients reported to date.
Actimab-A is the second product in clinical trials
Actinium Pharmaceuticals is developing a portfolio of radio-labelled antibodies that are particularly suited to the treatment of blood cancers, where their high specificity can target the widely disseminated individual cancerous cells effectively. Actimab-A is a second-generation product that uses lintuzumab (HuM195) to target the CD33 protein that is expressed predominantly on the surface of myeloid cells, which is linked to the α-emitting 225actinium construct. The Phase I element of the trial aims to determine the MTD, which will be used to determine the optimal dose for the Phase II portion of the trial.
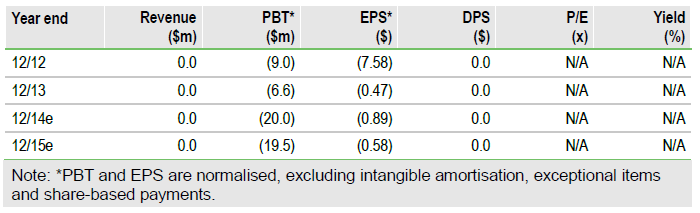
Valuation: rNPV model suggests $438m
Our valuation, based on an rNPV model, is $438m ($12.34 per undiluted share, $8.58 per diluted share). The rNPV is unchanged but the per share value reflects the new shares in issue following the February $20m fund-raise. The main value driver in our model is Iomab-B, the lead product that is Phase III ready, which we estimate could have peak sales of $2.9bn across various HSCT indications. The company has sufficient financial resources to progress this key program to a material value inflection point. Actimab-A is the second clinical product in the pipeline, with a further three programs in preclinical development.
To Read the Entire Report Please Click on the pdf File Below