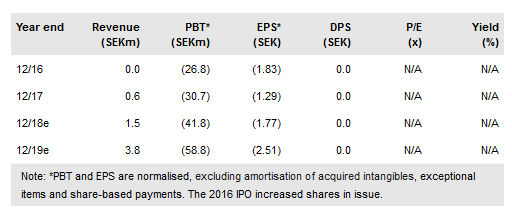
Acarix's (ST:ACARIX) annual report confirms reported 2017 revenues of SEK638k. Gross profit was SEK430k, with a gross margin rise to 75% in Q4. We do not expect any major sales upturn in 2018, as the key factor is German government reimbursement – this is not expected before 2019. There is additional sales potential in other European territories. We do not expect a US launch before 2022, but we have assumed a US trial starts in 2019. The indicative value remains at SEK448m (SEK19.46/share). Mr Lindholm is the interim CEO. Additional clinical studies are ongoing.
CAD-score – a clear indicator of low coronary risk
Acarix’s diagnostic device (CADScor) uses a highly sensitive microphone linked to a minimalist, self-contained processing module to record a patient’s diastolic heart sounds. There is a single-use patch. The CADScor V3 algorithm (validated by the Dan-NICAD clinical study) calculates a CAD-score rating. A negative test result was correct in 96% of tested patients, according to Winther et al (2017). The ongoing Dan-NICAD II study will enrol 2,000 patients with suspected stable coronary artery disease to add data, evaluate the test in patients aged 30–39 and should aid acceptance of CADScor among key opinion leaders. The planned ‘Seismo’ study with 200 patients aims to explore the use of CADScor for the early diagnosis of heart failure.
To read the entire report Please click on the pdf File Below: