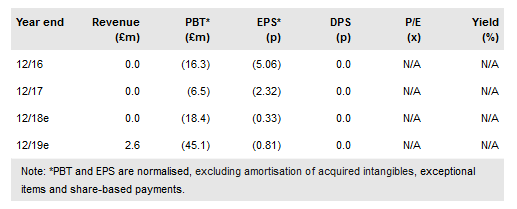
Acacia Pharma Group PLC (BR:ACPH) has resubmitted a New Drug Application (NDA) to the FDA for its lead asset BARHEMSYS (repurposed amisulpride) for the management of post-operative nausea and vomiting (PONV). While the original complete response letter (CRL) received was unexpected, the issues related to deficiencies at the contract manufacturers responsible for producing the active pharmaceutical ingredient (API) and not to any other part of the application or the API itself. Within 30 days of this resubmission, the FDA will determine whether the application constitutes a Class 1 (up to two months) or Class 2 (up to six months) review. Acacia maintains that a potential launch of BARHEMSYS for PONV rescue is possible in H119. It has reported a 9M18 net loss of £10.0m, driven by preparation activities for the commercial launch of BARHEMSYS. We value Acacia at €602m or €11.3/share.
BARHEMSYS launch still on track for H119
Since the CRL was received in October, Acacia and the API manufacturer have worked quickly to prepare a corrective and preventative action (CAPA) plan to address the outstanding deficiencies at the manufacturing facility. This plan has now been submitted to the FDA by the contract manufacturer alongside the NDA by Acacia. The time required by the FDA to review the NDA will depend on whether it is deemed a Class 1 or Class 2 resubmission. A launch under either scenario is still possible in H119. For launch, Acacia needs to build up its commercial operations, including the sizeable salesforce of 60-100 reps required for the PONV indication in the US. Funding in the near term will be required to build the required US organisation
To read the entire report Please click on the pdf File Below..