A player in the US prescription cough cold market
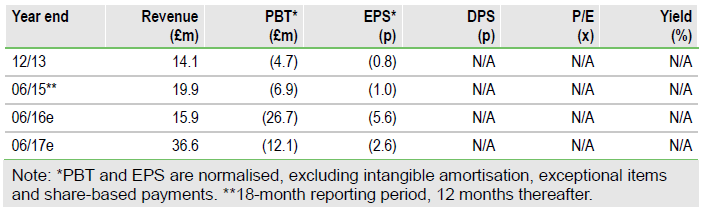
Vernalis (L:VER) is poised to complete the transition towards becoming a selfsustaining speciality pharma company, based on its five extended release cough cold products targeted at high-value prescribers. First out of the blocks is Tuzistra XR, launched in September. We forecast £5.8m cough cold sales in FY16 rising to £27.3m in FY17. Our DCF valuation is £439m.
Tuzistra XR – primary market segment worth $500m
Vernalis launched Tuzistra XR, an extended release (ER) or 12-hourly dosed cough cold treatment in September. The ER formulation offers greater convenience than immediate release products; there is only one other FDA-approved liquid ER prescription product owing to formulation complexities. Vernalis has attained tier 3 unrestricted formulary coverage for Tuzistra XR for over 120 million lives, which is to be a key driver of its ability to gain market share. Vernalis has a dedicated US salesforce in place ahead of the 2015/16 cough cold season.
To Read the Entire Report Please Click on the pdf File Below