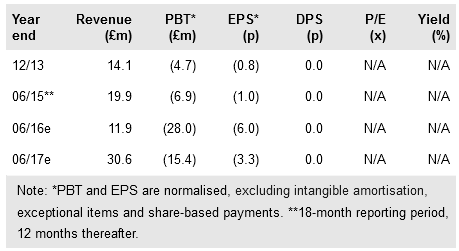
Vernalis (LON:VER) reported a modest £0.6m in Tuzistra XR sales over the first four months post-launch due to the mild cough cold season. The US launch of this prescription-only (Rx), extended release (ER) cough cold medicine is the first step in Vernalis’s transition into a commercial-stage speciality pharma company, targeting a $3.5bn market opportunity. The emphasis for year one of Tuzistra XR commercialisation is operational: establishing the platform for future sales growth. However, financial performance, in particular, during its second year on the market, will be an important determinant of whether Vernalis will need to raise new funds near term.
Solid operational progress offsets modest sales
Tuzistra XR’s slow launch into a mild season suggests FY16 consensus forecasts will fall despite a later peak than typical. So far, Vernalis has achieved an average net price per Rx of $70-75 based on a $177 wholesale acquisition cost per average 168ml Rx. Commercial focus is on improving physician awareness of and patient accessibility to Tuzistra XR. Improved reimbursement is vital: Tier 3 status has been achieved at c 55% of plans (vs 75-80% target); CVS Caremark (NYSE:CVS) is a major gap.
To read the entire report Please click on the pdf File Below