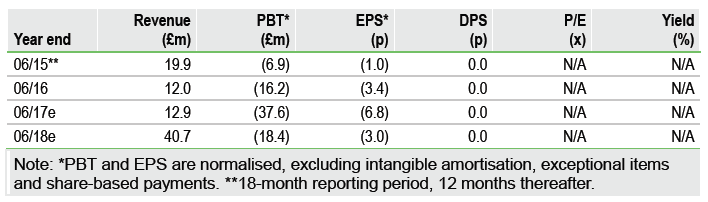
FDA acceptance of the CCP-08 NDA (setting a 4 August 2017 PDUFA date) 5 January 2017 advances Vernalis (LON:VER) towards its goal of building a speciality US franchise of extended release (ER) prescription-only (Rx) cough cold products. Potential 2017 approval of CCP-08 and CCP-07 (PDUFA date 20 April 2017) would enable launch into the 2017/18 cough cold season. Last reported cash of £78.6m (unaudited at end October) supports ongoing investment in operational initiatives to enhance Tuzistra XR sales growth this season and beyond. Successful execution will lay important foundations for the launches of CCP-07 and CCP-08.
Third cough cold programme under FDA review
CCP-08 is the third of Vernalis’s cough cold products to be accepted for NDA review; this triggers a milestone payment from Vernalis to partner Tris (we assume $3m). Potential CCP-07 and CCP-08 launches into the 2017/18 cough cold season will leverage the existing Tuzistra XR salesforce and coupled to higher revenue will drive Vernalis to profitability in FY19 on our forecasts. Two other pipeline assets (CCP-05 and CCP-06) are on track for achieving proof of concept in FY17.
To read the entire report Please click on the pdf File Below