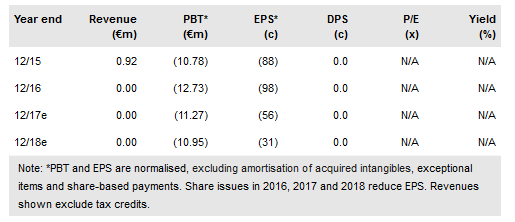
Txcell's (PA:TXCL) novel CAR-modified regulatory T-cell (CAR Treg) platform continues to develop well. TxCell has four indications in preclinical development with the first ever CAR Treg trial, in transplant rejection, anticipated by TxCell to start by late 2018. This could provide powerful clinical proof-of-concept data by 2020. In 2017, an €11.1m gross rights issue provided funding for 2017 with an operational cash use of €13m guided by management. In 2018, warrants could bring a further €10.8m in cash covering costs until the IND is filed for the first ever CAR Treg clinical trial. The indicative market cap remains at €74m.
Focus on CAR Treg with transplant in the lead
TxCell is now focused on the CAR Treg ENTrIA platform technology (see our last published note of 28 February 2017). ENTrIA uses chimeric antigen receptor (CAR) technology similar to that in the CAR T-cell cancer area; TxCell holds a core granted US patent. We expect ENTrIA to be an excellent basis for partnering and technology licensing – although deals are not shown in financial forecasts. The first clinical CAR Treg trial may start in 2018 for transplant, based on a key academic collaboration with published preclinical CAR Treg data. Other potential indications are lupus nephritis, bullous pemphigoid (skin) and multiple sclerosis. Ovasave, using ASTrIA technology with non-modified Tr1 Tregs is on hold.
To read the entire report Please click on the pdf File Below