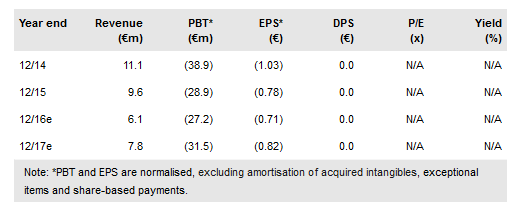
Transgene (PA:TRNG) has further expanded on its new strategy announced in January 2016 and clarified timelines for combination trials of its product candidates with immune checkpoint inhibitors. Six clinical trials are due to start before 2017 year end; its lead product candidates, Pexa-Vec (oncolytic virus) and TG4010 (therapeutic vaccine) in combination with Yervoy and Opdivo respectively should start phase II trials by year end. Restructuring in 2015 has significantly reduced costs and it is now financed through major inflection points in 2017. HY16 net cash of €33.4m includes short-term financial assets. We value Transgene at €4.40/share (€169.5m).
Pipeline focus
Transgene strategic focus is now on its therapeutic vaccines and oncolytic viruses in combination with other treatments, predominately immune checkpoint inhibitors (ICI). Its lead therapeutic vaccine product candidate, TG4010 is to initiate two trials in first- and second-line non-small cell lung cancer (NSCLC) in combination with ICIs. The first of which will start before year end in second-line treatment of NSCLC alongside Opdivo (Bristol-Myers Squibb, BMY). Transgene plans to start six trials across all product candidates in the next 3-18 months, five of which are in combination with ICIs.
To read the entire report Please click on the pdf File Below