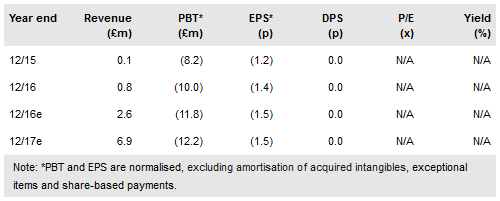
Tissue Regenix's (LON:TRX) interim results are in line with our expectations and demonstrate the progress the company is making to achieve various milestones in each of its divisions. In particular, its Wound Care division made important progress in its commercialisation and achieved 510k market clearance for its SurgiPure XD product and increased its Medicare coverage for DermaPure (to 93%). We have increased our valuation to £364m (previously £338m) due to an updated exchange rate (£/$1.22), rebasing the model and using an updated H116 cash position of £13.5m.
Commercialisation progress
DermaPure has continued to perform well, with H1 revenue of £631k. This is an increase on the previous year (£252k), following an increase in Medicare coverage (currently 10 out of 11 reimbursement agencies), an expanded portfolio and increased distributorship. The company indicates that H2 should follow a similar revenue profile as previous years, with acceleration in sales towards the year end. Progress in H1 included the SurgiPure XD 510k market clearance in the US. This was important as it not only enables the launch of SurgiPure XD to the US market (expected in H217), but also validated the dCELL safety and efficacy data, important for future development of the technology in the US market.